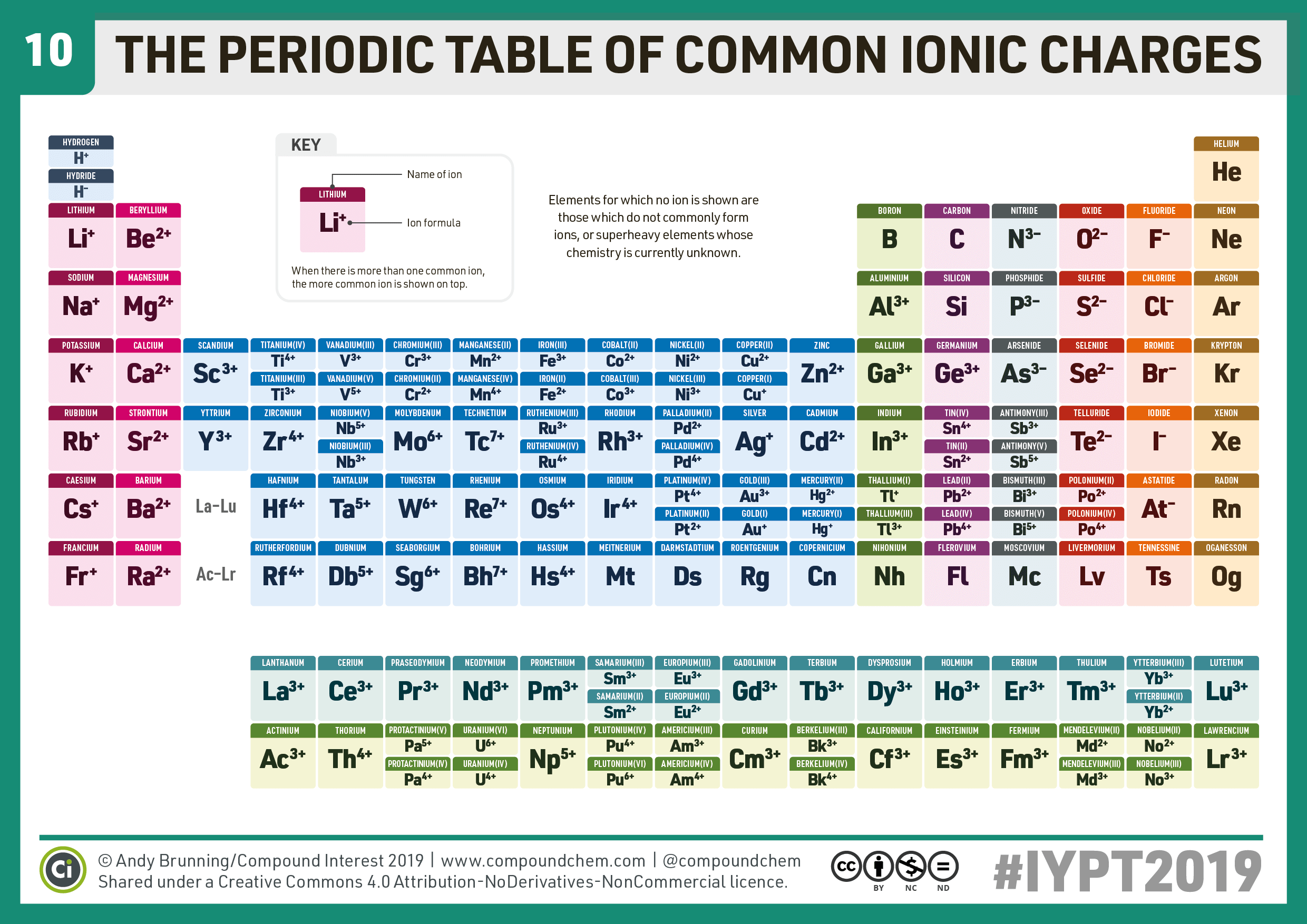 
Finally, we should comprehend exchange resins and ionic radii trends. We should also be able to understand which ions will gain or lose electrons. We should be comfortable identifying ions, cations, and anions. Now that we’ve taken a look at some examples of cations and anions through an ionic compound reaction. We will use the same ions shown in our writing ions graphic above.įigure 6: Examples of ion transfer shown in an ionic compound reaction where Sodium Chloride and Magnesium Oxide are produced. We can also use Lewis dot diagrams to show electron transfer in ionic compounds, which is precisely what we are going to do now. It is recognized as a ferromagnetic element. The density of cobalt is 8.9 grams per cubic centimeter. Simplified illustrations of a molecule's valence electrons are known as Lewis dot diagrams. Cobalt has an atomic number of 27 and is a lustrous silvery-blue metal.It has a melting point of 1495 ☌ (2723 ☏) and a boiling point of 2927 ☌ (5301 ☏). To achieve this, we will use Lewis Diagrams. The next step is to look at how ionic compounds form. Now that we have learned about which elements tend to create cations and which ones tend to create anions. It’s insoluble and contains microbeads that are porous enough to trap specific ions, according to charge, facilitating the process known as ion exchange.įor more detailed information regarding valence electrons or Lewis Diagrams, please reference our “Valence Electrons” or “Lewis Diagrams” articles. For example, group 17 elements (one group left of the noble gases) form 1 ions group 16 elements (two groups left) form 2 ions, and so on. Resin is a highly viscous substance, often made using plants. Moving from the far right to the left on the periodic table, elements often form anions with a negative charge equal to the number of groups moved left from the noble gases. Cation and Anion Ion Exchange ResinĮarlier in the article, we mentioned that certain substances can act as media for ion exchange. Write chemical formulas for the ionic compounds made from each set of ions. In other words, cations have a smaller ionic radius, while anions have a larger ionic radius when compared to their element’s respective atomic radius. Cobalt is a necessary component of vitamin B 12 (hydroxocobalamin) and a fundamental coenzyme of cell mitosis. Less electron repulsion results in a smaller ionic radius. In the human body this element is present in amounts from 1 to 2 mg: we can find it in the heart, liver, kidney, and spleen, and considerably smaller quantities in the pancreas, brain, and serum 10, 11. The opposite occurs with cations, which result from the loss of electrons. This increase in electron repulsion pushes electrons further apart, resulting in a larger ionic radius. The size differences in radii arise because as neutral atoms gain electrons and become anions, more electrons occupy the outer orbitals, leading to increased electron repulsion. In comparison, cations have a smaller ionic radius when compared to the same element’s atomic radius.Ĭonfused? It’s all right! The illustration below gives a visual representation of radial size differences.įigure 3: Cations and anions radius compared to their element’s respective atomic radius. Carbonate, CO32-, Carbonic acid, H2CO3 (unstable, decomposes into H2O and CO2). Organic carbonyls can also bind to metal atoms or ions.For more detailed information regarding periodic trends, please reference our “Periodic Trends” or “Periodic Trends: General Trends” articles.Īnions have a greater ionic radius when compared to the same element’s atomic radius. Name and formula of the acid containing the anion.The resulting complexes are important because the carbonyl becomes "activated" or ready to accept nucleophiles. The oxygen lone pair donates to the metal atom or ion. Normally, we think of them as simple lone pair donors. These compounds bind to transition metals in a very different way than carbon monoxide. "Organic" carbonyls, such as aldehydes and ketones, can also bind to transition metals, as you may have seen before. explain why the terminal carbonyls display stretching frequencies of 20 cm -1, but the bridging carbonyls display a stretching frequency of 1829 cm -1.This complex also features a metal-metal bond. For example, the iron cluster Fe2(CO)9 contains six "terminal" carbonyls, bound to only one iron each, and three "bridging" carbonyls, each of which is bound to both iron atoms. Sometimes, carbonyls can bridge between two metals. \): Donation from metal to CO in qualitative molecular orbital terms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
